Am Fam Physician. 2022;105(4):350-352
Author disclosure: No relevant financial relationships.
Clinical practice guidelines, essential tools for evidence-based practice, have proliferated over the past few decades but remain controversial for several reasons, including discordance among guidelines, the influence of intellectual and financial conflicts of interest, and a lack of adherence to standards for developing trustworthy guidelines. In this editorial, we will use recent hypertension guidelines to illustrate guideline discordance and then propose how to identify trustworthy guidelines.
Comprehensive, systematic evidence search
Evidence linked directly to recommendations and a strength of recommendation
Emphasis on patient-oriented rather than disease-oriented outcomes
Transparency
Minimal conflicts of interest
Prospective validation
Recommendations that offer flexibility in various clinical situations
Multiple organizations have issued guidelines on managing patients with hypertension. A recent analysis identified eight guidelines, released between 2010 and 2018, with 71 separate recommendations.4 However, three recommendations were unique to a single guideline, leaving 68 for the analysis. The recommendations included variations on blood pressure measurement methods, laboratory and other test indications, and treatment thresholds. Each recommendation was rated on the direction (e.g., do XYZ or do not do XYZ) and the overall strength. Twenty-two (32%) of the recommendations were concordant in both the direction and strength. However, 28 (41%) of the recommendations had inconsistent directions. For example, the guidelines recommended varying treatment targets (e.g., target blood pressure less than 150/90, 140/90, or 130/80 mm Hg, or systolic pressure less than 120 mm Hg) and varying preferred initial therapies (e.g., thiazides, angiotensin-converting enzyme inhibitors, calcium channel blockers, beta blockers). Sensitivity analyses to determine the role of the underlying strength of ratings, source of recommendations, and importance of recommendations failed to reveal any causes for guideline inconsistency. We suggest that several guideline development factors not addressed by the authors are likely explanations:
Interpretation: The developers of the guidelines value some outcomes more than others.
Speculation: When evidence is lacking, experts provide their best guess.
Extrapolation: Recommendations extrapolate beyond the research, whether to specific interventions (e.g., blood pressure targets) or to the study populations.
Representation: The panel of developers lacks relevant stakeholders (e.g., patients, primary care physicians).
Oversimplification: Guidelines apply a one-size-fits-all approach that ignores nuances about disease severity, patient comorbidity, age, and other contextual factors.
Overcomplication: Guidelines address too many specific questions.
Application: Guidelines do not consistently address feasibility and lack clarity on whether they apply to individuals or systems.
Money: Developers have conflicts of interest, or they do not address the costs of guideline implementation.
The true measure of a guideline is whether clinicians who use and follow it have improved patient outcomes, preferably in the setting of a randomized trial. However, prospective validation of guidelines rarely occurs. Guidelines resulting in worse patient outcomes compared with other guidelines or watchful waiting should be retired. For example, a registry-based study found that the use of anticoagulant therapy for atrial fibrillation based on guidelines from the European Society of Cardiology was associated with a nearly three-fold increased risk of hemorrhage compared with treatment based on guidelines from the American College of Chest Physicians, which did not recommend anticoagulant therapy for older people with a CHADS2 (congestive heart failure, hypertension, age, diabetes, stroke) score of 0.5
What should clinicians do? First, they should be skeptical. Next, they should identify a trusted source of guidelines that follow good practices. For example, a collection of Practice Guidelines published in American Family Physician is available at https://www.aafp.org/afp/practguide. The Emergency Care Research Institute Guidelines Trust (https://guidelines.ecri.org/) is a non-profit entity that provides a repository of guidelines accompanied by a scorecard that grades the guidelines against the standards developed by the National Academy of Medicine, which are described in this Editorial. Although this resource is free, it requires registration, and it is unclear how often the guidelines are updated.
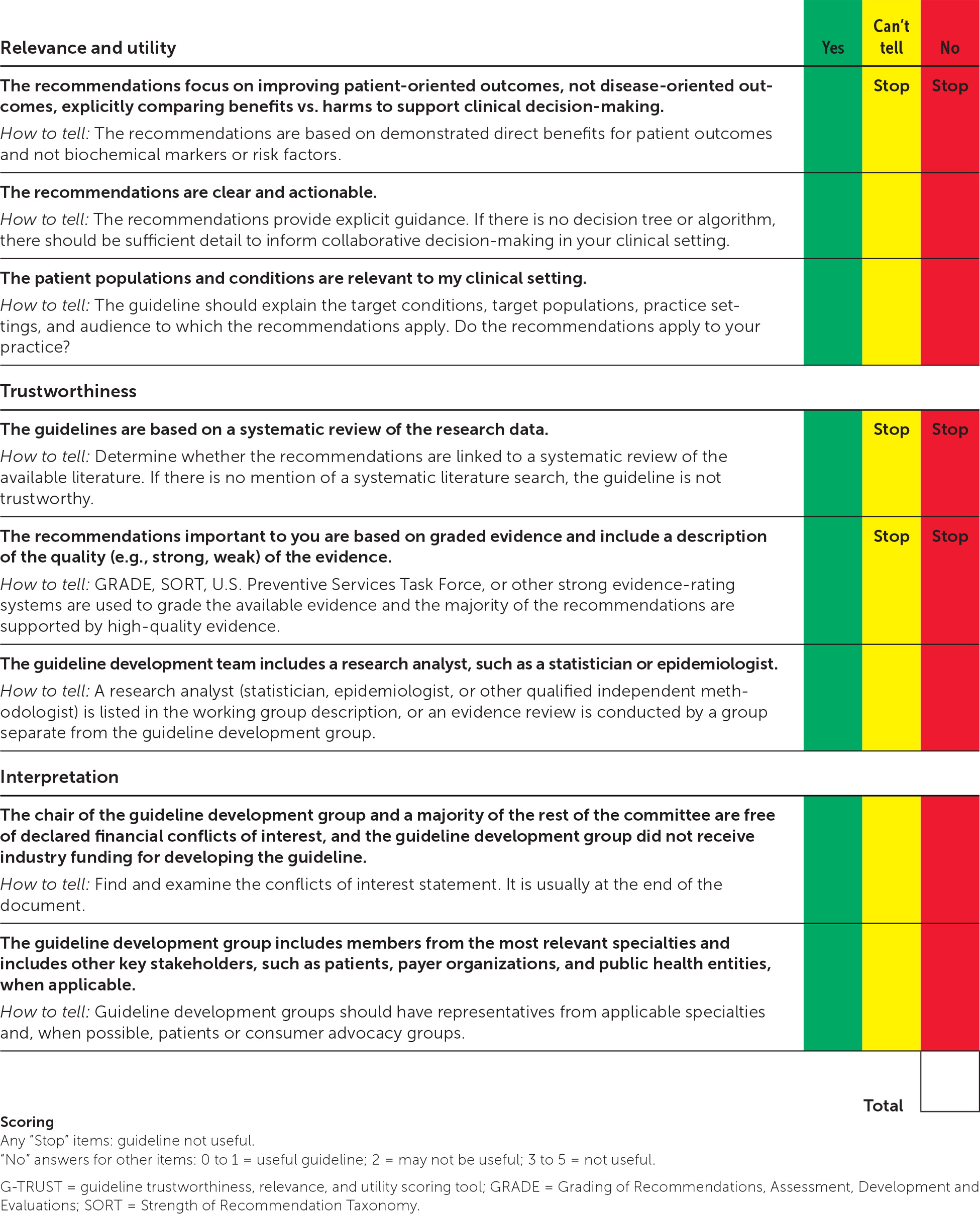
A more rigorous proposition is that clinicians grade guidelines themselves using the Guideline Trustworthiness, Relevance, and Utility Scoring Tool (G-TRUST)6 or a similar tool. The G-TRUST (Figure 1) is a simple tool for clinicians to independently assess the trustworthiness and utility of guidelines. Although its validation has been limited, its face validity is high because it prioritizes patient-oriented outcomes and adherence to good guideline development processes. Finally, clinicians need to be prepared to stop the use of guidelines found to be ineffective or harmful.
Sir Muir Gray of the United Kingdom National Screening Committee famously stated, “All screening programs do harm; some do good as well.” This is also true for clinical practice guidelines. To do more good than harm to patients, guideline panels must at a minimum use transparent and inclusive processes and eliminate the process factors that result in discordant and potentially biased guidelines.