
Am Fam Physician. 1999;59(9):2487-2500
A more recent article on intrapartum fetal monitoring is available.
Electronic fetal heart rate monitoring is commonly used to assess fetal well-being during labor. Although detection of fetal compromise is one benefit of fetal monitoring, there are also risks, including false-positive tests that may result in unnecessary surgical intervention. Since variable and inconsistent interpretation of fetal heart rate tracings may affect management, a systematic approach to interpreting the patterns is important. The fetal heart rate undergoes constant and minute adjustments in response to the fetal environment and stimuli. Fetal heart rate patterns are classified as reassuring, nonreassuring or ominous. Nonreassuring patterns such as fetal tachycardia, bradycardia and late decelerations with good short-term variability require intervention to rule out fetal acidosis. Ominous patterns require emergency intrauterine fetal resuscitation and immediate delivery. Differentiating between a reassuring and nonreassuring fetal heart rate pattern is the essence of accurate interpretation, which is essential to guide appropriate triage decisions.
Electronic fetal heart rate monitoring (EFM) was first introduced at Yale University in 1958.1 Since then, continuous EFM has been widely used in the detection of fetal compromise and the assessment of the influence of the intrauterine environment on fetal welfare.
In 1991, the National Center for Health Statistics reported that EFM was used in 755 cases per 1,000 live births in the United States.2 In many hospitals, it is routinely used during labor, especially in high-risk patients.
Monitoring the Fetal Heart Rate
Auscultation of the fetal heart rate (FHR) is performed by external or internal means. External monitoring is performed using a hand-held Doppler ultrasound probe to auscultate and count the FHR during a uterine contraction and for 30 seconds thereafter to identify fetal response. It may also be performed using an external transducer, which is placed on the maternal abdomen and held in place by an elastic belt or girdle. The transducer uses Doppler ultrasound to detect fetal heart motion and is connected to an FHR monitor. The monitor calculates and records the FHR on a continuous strip of paper. Recently, second-generation fetal monitors have incorporated microprocessors and mathematic procedures to improve the FHR signal and the accuracy of the recording.3 Internal monitoring is performed by attaching a screw-type electrode to the fetal scalp with a connection to an FHR monitor. The fetal membranes must be ruptured, and the cervix must be at least partially dilated before the electrode may be placed on the fetal scalp.
The American College of Obstetricians and Gynecologists (ACOG) states that with specific intervals, intermittent auscultation of the FHR is equivalent to continuous EFM in detecting fetal compromise.4 ACOG has recommended a 1:1 nurse-patient ratio if intermittent auscultation is used as the primary technique of FHR surveillance.4 The recommended intermittent auscultation protocol calls for auscultation every 30 minutes for low-risk patients in the active phase of labor and every 15 minutes in the second stage of labor.4 Continuous EFM is indicated when abnormalities occur with intermittent auscultation and for use in high-risk patients. Table 1 lists examples of the criteria that have been used to categorize patients as high risk.
Benefits and Risks of EFM
One benefit of EFM is to detect early fetal distress resulting from fetal hypoxia and metabolic acidosis. In the United States, an estimated 700 infant deaths per year are associated with intrauterine hypoxia and birth asphyxia.5 Another benefit of EFM includes closer assessment of high-risk mothers. Most patients who undergo internal fetal monitoring during labor accept monitoring as a positive experience.6
The most important risk of EFM is its tendency to produce false-positive results. Unfortunately, precise information about the frequency of false-positive results is lacking, and this lack is due in large part to the absence of accepted definitions of fetal distress.7 Meta-analysis of all published randomized trials has shown that EFM is associated with increased rates of surgical intervention resulting in increased costs.8 These results show that 38 extra cesarean deliveries and 30 extra forceps operations are performed per 1,000 births with continuous EFM versus intermittent auscultation. Variable and inconsistent interpretation of tracings by clinicians may affect management of patients. The effect of continuous EFM monitoring on malpractice liability has not been well established. Other rare risks associated with EFM include fetal scalp infection and uterine perforation with the intra-uterine tocometer or catheter.
Some clinicians have argued that this unproven technology has become the standard for all patients designated high risk and has been widely applied to low-risk patients as well.9 The worldwide acceptance of EFM reflects a confidence in the importance of electronic monitoring and concerns about the applicability of auscultation.10 However, in a 1996 report, the U.S. Preventive Services Task Force7 did not recommend the use of routine EFM in low-risk women in labor.
Interpreting FHR Patterns
A systematic approach is recommended when reading FHR recordings to avoid misinterpretation (Table 2). The FHR recordings may be interpreted as reassuring, nonreassuring or ominous, according to the pattern of the tracing. Reassuring patterns correlate well with a good fetal outcome, while nonreassuring patterns do not. Evaluation of fetal well-being using fetal scalp stimulation, pH measurement, or both, is recommended for use in patients with nonreassuring patterns.11,12 Evaluation for immediate delivery is recommended for patients with ominous patterns. Table 3 lists examples of nonreassuring and ominous patterns. The FHR tracing should be interpreted only in the context of the clinical scenario, and any therapeutic intervention should consider the maternal condition as well as that of the fetus. For example, fetuses with intrauterine growth restriction are unusually susceptible to the effect of hypoxemia, which tends to progress rapidly.4
| 1. Evaluate recording—is it continuous and adequate for interpretation? |
| 2. Identify type of monitor used—external versus internal, first-generation versus second-generation. |
| 3. Identify baseline fetal heart rate and presence of variability, both long-term and beat-to-beat (short-term). |
| 4. Determine whether accelerations or decelerations from the baseline occur. |
| 5. Identify pattern of uterine contractions, including regularity, rate, intensity, duration and baseline tone between contractions. |
| 6. Correlate accelerations and decelerations with uterine contractions and identify the pattern. |
| 7. Identify changes in the FHR recording over time, if possible. |
| 8. Conclude whether the FHR recording is reassuring, nonreassuring or ominous. |
| 9. Develop a plan, in the context of the clinical scenario, according to interpretation of the FHR. |
| 10. Document in detail interpretation of FHR, clinical conclusion and plan of management. |
| Nonreassuring patterns |
| Fetal tachycardia |
| Fetal bradycardia |
| Saltatory variability |
| Variable decelerations associated with a nonreassuring pattern |
| Late decelerations with preserved beat-to-beat variability |
| Ominous patterns |
| Persistent late decelerations with loss of beat-to-beat variability |
| Nonreassuring variable decelerations associated with loss of beat-to-beat variability |
| Prolonged severe bradycardia |
| Sinusoidal pattern |
| Confirmed loss of beat-to-beat variability not associated with fetal quiescence, medications or severe prematurity |
A growing body of evidence suggests that, when properly interpreted, FHR assessment may be equal or superior to measurement of fetal blood pH in the prediction of both good and bad fetal outcomes.13 Fetuses with a normal pH, i.e., greater than 7.25, respond with an acceleration of the fetal heart rate following fetal scalp stimulation. Fetal scalp sampling for pH is recommended if there is no acceleration with scalp stimulation.11
A scalp pH less than 7.25 but greater than 7.20 is considered suspicious or borderline. Results in this range must also be interpreted in light of the FHR pattern and the progress of labor, and generally should be repeated after 15 to 30 minutes. A scalp pH of less than 7.20 is considered abnormal and generally is an indication for intervention, immediate delivery, or both.12 A pH less than 7.20 should also be assumed in the absence of an acceleration following fetal scalp stimulation when fetal scalp pH sampling is not available. Table 4 lists recommended emergency interventions for nonreassuring patterns.4,14 These interventions should also be considered for ominous patterns while preparations for immediate delivery are initiated.
| Call for assistance |
| Administer oxygen through a tight-fitting face mask |
| Change maternal position (lateral or knee-chest) |
| Administer fluid bolus (lactated Ringer's solution) |
| Perform a vaginal examination and fetal scalp stimulation |
| When possible, determine and correct the cause of the pattern |
| Consider tocolysis (for uterine tetany or hyperstimulation) |
| Discontinue oxytocin if used |
| Consider amnioinfusion (for variable decelerations) |
| Determine whether operative intervention is warranted and, if so, how urgently it is needed |
FHR Patterns
BASELINE FHR
The FHR is controlled by the autonomic nervous system. The inhibitory influence on the heart rate is conveyed by the vagus nerve, whereas excitatory influence is conveyed by the sympathetic nervous system. Progressive vagal dominance occurs as the fetus approaches term and, after birth, results in a gradual decrease in the baseline FHR. Stimulation of the peripheral nerves of the fetus by its own activity (such as movement) or by uterine contractions causes acceleration of the FHR.15
Baroreceptors influence the FHR through the vagus nerve in response to change in fetal blood pressure. Almost any stressful situation in the fetus evokes the baroreceptor reflex, which elicits selective peripheral vasoconstriction and hypertension with a resultant bradycardia. Hypoxia, uterine contractions, fetal head compression and perhaps fetal grunting or defecation result in a similar response.
Chemoreceptors located in the aortic and carotid bodies respond to hypoxia, excess carbon dioxide and acidosis, producing tachycardia and hypertension.15 The FHR is under constant and minute adjustment in response to the constant changes in the fetal environment and external stimuli.
The normal FHR range is between 120 and 160 beats per minute (bpm). The baseline rate is interpreted as changed if the alteration persists for more than 15 minutes. Prematurity, maternal anxiety and maternal fever may increase the baseline rate, while fetal maturity decreases the baseline rate.
FHR VARIABILITY
The FHR is under constant variation from the baseline (Figure 1). This variability reflects a healthy nervous system, chemoreceptors, baroreceptors and cardiac responsiveness. Prematurity decreases variability16; therefore, there is little rate fluctuation before 28 weeks. Variability should be normal after 32 weeks.17 Fetal hypoxia, congenital heart anomalies and fetal tachycardia also cause decreased variability. Beat-to-beat or short-term variability is the oscillation of the FHR around the baseline in amplitude of 5 to 10 bpm. Long-term variability is a somewhat slower oscillation in heart rate and has a frequency of three to 10 cycles per minute and an amplitude of 10 to 25 bpm. Clinically, loss of beat-to-beat variability is more significant than loss of long-term variability and may be ominous.18 Decreased or absent variability should generally be confirmed by fetal scalp electrode monitoring when possible.
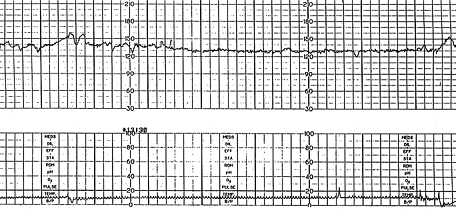
Interpretation of the FHR variability from an external tracing appears to be more reliable when a second-generation fetal monitor is used than when a first-generation monitor is used.3 Loss of variability may be uncomplicated and may be the result of fetal quiescence (rest-activity cycle or behavior state), in which case the variability usually increases spontaneously within 30 to 40 minutes.19 Uncomplicated loss of variability may also be caused by central nervous system depressants such as morphine, diazepam (Valium) and magnesium sulfate; parasympatholytic agents such as atropine and hydroxyzine (Atarax); and centrally acting adrenergic agents such as methyldopa (Aldomet), in clinical dosages.19
Beta-adrenergic agonists used to inhibit labor, such as ritodrine (Yutopar) and terbutaline (Bricanyl), may cause a decrease in variability only if given at dosage levels sufficient to raise the fetal heart rate above 160 bpm.19 Uncomplicated loss of variability usually signifies no risk or a minimally increased risk of acidosis19,20 or low Apgar scores.21 Decreased FHR variability in combination with late or variable deceleration patterns indicates an increased risk of fetal preacidosis (pH 7.20 to 7.25) or acidosis (pH less than 7.20)19,20,22 and signifies that the infant will be depressed at birth.21 The combination of late or severe variable decelerations with loss of variability is particularly ominous.19 The occurrence of a late or worsening variable deceleration pattern in the presence of normal variability generally means that the fetal stress is either of a mild degree or of recent origin19; however, this pattern is considered nonreassuring.
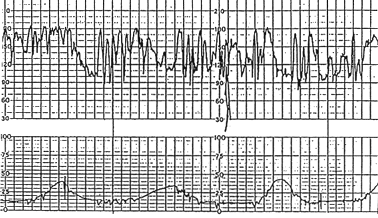
Increased variability in the baseline FHR is present when the oscillations exceed 25 bpm (Figure 2). This pattern is sometimes called a saltatory pattern and is usually caused by acute hypoxia or mechanical compression of the umbilical cord. This pattern is most often seen during the second stage of labor. The presence of a saltatory pattern, especially when paired with decelerations, should warn the physician to look for and try to correct possible causes of acute hypoxia and to be alert for signs that the hypoxia is progressing to acidosis.21 Although it is a nonreassuring pattern, the saltatory pattern is usually not an indication for immediate delivery.19
FETAL TACHYCARDIA
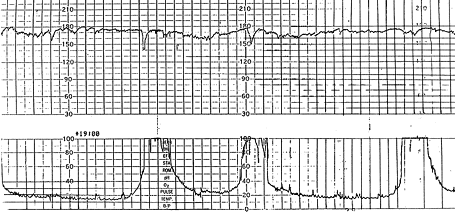
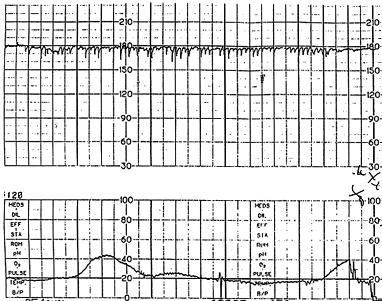
Fetal tachycardia is defined as a baseline heart rate greater than 160 bpm and is considered a nonreassuring pattern (Figure 3). Tachycardia is considered mild when the heart rate is 160 to 180 bpm and severe when greater than 180 bpm. Tachycardia greater than 200 bpm is usually due to fetal tachyarrhythmia (Figure 4) or congenital anomalies rather than hypoxia alone.16 Causes of fetal tachycardia are listed in Table 5.
| Fetal hypoxia | |
| Maternal fever | |
| Hyperthyroidism | |
| Maternal or fetal anemia | |
| Parasympatholytic drugs | |
| Atropine | |
| Hydroxyzine (Atarax) | |
| Sympathomimetic drugs | |
| Ritodrine (Yutopar) | |
| Terbutaline (Bricanyl) | |
| Chorioamnionitis | |
| Fetal tachyarrhythmia | |
| Prematurity | |
Persistent tachycardia greater than 180 bpm, especially when it occurs in conjunction with maternal fever, suggests chorioamnionitis. Fetal tachycardia may be a sign of increased fetal stress when it persists for 10 minutes or longer, but it is usually not associated with severe fetal distress unless decreased variability or another abnormality is present.4,11,17
FETAL BRADYCARDIA
Fetal bradycardia is defined as a baseline heart rate less than 120 bpm. Bradycardia in the range of 100 to 120 bpm with normal variability is not associated with fetal acidosis. Bradycardia of this degree is common in post-date gestations and in fetuses with occiput posterior or transverse presentations.16 Bradycardia less than 100 bpm occurs in fetuses with congenital heart abnormalities or myocardial conduction defects, such as those occurring in conjunction with maternal collagen vascular disease.16 Moderate bradycardia of 80 to 100 bpm is a nonreassuring pattern. Severe prolonged bradycardia of less than 80 bpm that lasts for three minutes or longer is an ominous finding indicating severe hypoxia and is often a terminal event.4,11,16 Causes of prolonged severe bradycardia are listed in Table 6. If the cause cannot be identified and corrected, immediate delivery is recommended.
| Prolonged cord compression |
| Cord prolapse |
| Tetanic uterine contractions |
| Paracervical block |
| Epidural and spinal anesthesia |
| Maternal seizures |
| Rapid descent |
| Vigorous vaginal examination |
Periodic FHR Changes
ACCELERATIONS
Accelerations are transient increases in the FHR (Figure 1). They are usually associated with fetal movement, vaginal examinations, uterine contractions, umbilical vein compression, fetal scalp stimulation or even external acoustic stimulation.15 The presence of accelerations is considered a reassuring sign of fetal well-being. An acceleration pattern preceding or following a variable deceleration (the “shoulders” of the deceleration) is seen only when the fetus is not hypoxic.15 Accelerations are the basis for the nonstress test (NST). The presence of at least two accelerations, each lasting for 15 or more seconds above baseline and peaking at 15 or more bpm, in a 20-minute period is considered a reactive NST.
EARLY DECELERATIONS
Early decelerations are caused by fetal head compression during uterine contraction, resulting in vagal stimulation and slowing of the heart rate. This type of deceleration has a uniform shape, with a slow onset that coincides with the start of the contraction and a slow return to the baseline that coincides with the end of the contraction. Thus, it has the characteristic mirror image of the contraction (Figure 5). Although these decelerations are not associated with fetal distress and thus are reassuring, they must be carefully differentiated from the other, nonreassuring decelerations.

LATE DECELERATIONS
Late decelerations are associated with uteroplacental insufficiency and are provoked by uterine contractions. Any decrease in uterine blood flow or placental dysfunction can cause late decelerations. Maternal hypotension and uterine hyperstimulation may decrease uterine blood flow. Postdate gestation, preeclampsia, chronic hypertension and diabetes mellitus are among the causes of placental dysfunction. Other maternal conditions such as acidosis and hypovolemia associated with diabetic ketoacidosis may lead to a decrease in uterine blood flow, late decelerations and decreased baseline variability.23
A late deceleration is a symmetric fall in the fetal heart rate, beginning at or after the peak of the uterine contraction and returning to baseline only after the contraction has ended (Figure 6). The descent and return are gradual and smooth. Regardless of the depth of the deceleration, all late decelerations are considered potentially ominous. A pattern of persistent late decelerations is nonreassuring, and further evaluation of the fetal pH is indicated.16 Persistent late decelerations associated with decreased beat-to-beat variability is an ominous pattern19 (Figure 7).


VARIABLE DECELERATIONS
Variable decelerations are shown by an acute fall in the FHR with a rapid downslope and a variable recovery phase. They are characteristically variable in duration, intensity and timing. They resemble the letter “U,” “V” or “W” and may not bear a constant relationship to uterine contractions. They are the most commonly encountered patterns during labor and occur frequently in patients who have experienced premature rupture of membranes17 and decreased amniotic fluid volume.24 Variable decelerations are caused by compression of the umbilical cord. Pressure on the cord initially occludes the umbilical vein, which results in an acceleration (the shoulder of the deceleration) and indicates a healthy response. This is followed by occlusion of the umbilical artery, which results in the sharp downslope. Finally, the recovery phase is due to the relief of the compression and the sharp return to the baseline, which may be followed by another healthy brief acceleration or shoulder (Figure 8).

Variable decelerations may be classified according to their depth and duration as mild, when the depth is above 80 bpm and the duration is less than 30 seconds; moderate, when the depth is between 70 and 80 bpm and the duration is between 30 and 60 seconds; and severe, when the depth is below 70 bpm and the duration is longer than 60 seconds.4,11,24 Variable decelerations are generally associated with a favorable outcome.25 However, a persistent variable deceleration pattern, if not corrected, may lead to acidosis and fetal distress24 and therefore is nonreassuring. Table 7 lists signs associated with variable decelerations indicating hypoxemia4,11,26 (Figures 9 and 10). Nonreassuring variable decelerations associated with the loss of beat-to-beat variability correlate substantially with fetal acidosis4 and therefore represent an ominous pattern.
| Increased severity of the deceleration |
| Late onset and gradual return phase |
| Loss of “shoulders” on FHR recording |
| A blunt acceleration or “overshoot” after severe deceleration26 (Figure 9) |
| Unexplained tachycardia |
| Saltatory variability |
| Late decelerations or late return to baseline (Figure 10) |
| Decreased variability |

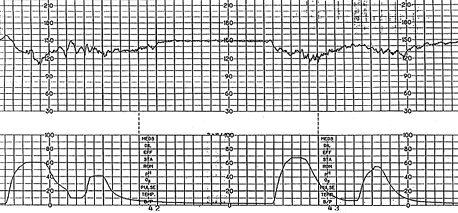
SINUSOIDAL PATTERN
The true sinusoidal pattern is rare but ominous and is associated with high rates of fetal morbidity and mortality.24 It is a regular, smooth, undulating form typical of a sine wave that occurs with a frequency of two to five cycles per minute and an amplitude range of five to 15 bpm. It is also characterized by a stable baseline heart rate of 120 to 160 bpm and absent beat-to-beat variability. It indicates severe fetal anemia, as occurs in cases of Rh disease or severe hypoxia.24 It should be differentiated from the “pseudosinusoidal” pattern (Figure 11a), which is a benign, uniform long-term variability pattern. A pseudosinusoidal pattern shows less regularity in the shape and amplitude of the variability waves and the presence of beat-to-beat variability, compared with the true sinusoidal pattern (Figure 11b).
